Research
Our research investigates how viruses reprogram host cell organelles to support replication and evade immune responses. We focus on lipid droplets (LDs), dynamic organelles central to lipid metabolism, inflammation, and innate immunity, which are increasingly recognized as key platforms for virus–host interactions.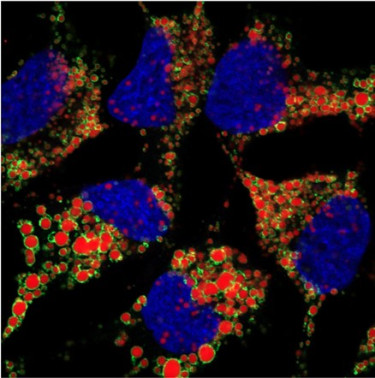
Using positive-strand RNA viruses, particularly coronaviruses, we study how pathogens exploit lipid droplets and their contact sites with organelles such as the endoplasmic reticulum and mitochondria to promote replication. We also explore how the host reshapes lipid droplets to counter infection, engaging innate immune signaling and organelle-based antiviral defenses.
A central objective is to define the LD proteome during viral infection, identifying viral and host proteins that localize to LDs, delineating their roles in replication and immune control, and uncovering how these interactions shape infection outcome and disease severity. To translate mechanistic insights into therapeutic strategies, we aim to evaluate the functional relevance of candidate proteins and pathways in preclinical models.
In parallel, we assess the efficacy of novel antiviral compounds and vaccine candidates. By integrating cell biology, omics technologies, and translational virology, our work aims to advance the molecular understanding of virus–organelle interactions and inform the development of targeted interventions for viral control and pandemic preparedness.
Main research questions:
- What roles do viral and host proteins play on lipid droplets and their contact sites during infection, and how do these interactions influence viral replication and host immune responses?
- Can lipid droplets and LD-associated pathways be leveraged as targets for antiviral therapies?
- How do in vivo models reveal the dynamics of antiviral innate immunity and the contribution of virus-induced organelle remodeling?
- How effective are candidate antiviral agents and vaccines in physiologically relevant preclinical models?
Approaches:
- In vitro systems and murine models
- Molecular virology and genetic approaches.
- Advanced imaging using confocal fluorescence and electron microscopy
- Quantitative proteomics and lipidomics via mass spectrometry
- Preclinical antiviral drug and vaccine testing
Group members
- Dr. Sebenzile K. Myeni
- Erin van der Venne (PhD student)
- Marissa E. Linger (Research technician)
- Sytze H.T. Jorritsma (Research technician)
- Sanne van Velzen (Research technician)
- Irene de Graaf (Master student)
Partners / Collaborators
We collaborate across LUMC and beyond, contributing to multiple national and international research consortia. Key partners include: Eric J. Snijder (LUCID), Bruno Guigas (LUCID), Bart Everts (LUCID), Peter van Veelen (CPM), Martin Giera (CPM), Igor Sidorov (LUCID), Martijn van Hemert (LUCID), Montserrat Barcena (CCB), Ramon Arens (IMMU), CARE Consortium partners, PANVIPREP Consortium partners.
Publications
Myeni, S.K., Bredenbeek, P.J., Knaap, R.C.M. et al. Engineering potent live attenuated coronavirus vaccines by targeted inactivation of the immune evasive viral deubiquitinase. Nat Commun 14, 1141 (2023). https://doi.org/10.1038/s41467-023-36754-z
van Huizen M, Bloeme-Ter Horst JR, de Gruyter HLM, Geurink PP, van der Heden van Noort GJ, Knaap RCM, Nelemans T, Ogando NS, Leijs AA, Urakova N, Mark BL, Snijder EJ, Kikkert M, Myeni SK. Deubiquitinating activity of SARS-CoV-2 papain-like protease does not influence virus replication or innate immune responses in vivo. PLoS Pathog. (2024.https://doi.org/10.1371/journal.ppat.1012100
van Huizen M, Vendrell XM, de Gruyter HLM, Boomaars-van der Zanden AL, van der Meer Y, Snijder EJ, Kikkert M, Myeni SK. The Main Protease of Middle East Respiratory Syndrome Coronavirus Induces Cleavage of Mitochondrial Antiviral Signaling Protein to Antagonize the Innate Immune Response. Viruses. (2024). https://doi.org/10.3390/v16020256
Myeni SK, Leijs AA, Bredenbeek PJ, Morales ST, Linger ME, Fougeroux C, van Zanen-Gerhardt S, Zander SAL, Sander AF, Kikkert M. Protection of K18-hACE2 Mice against SARS-CoV-2 Challenge by a Capsid Virus-like Particle-Based Vaccine. Vaccines. (2024). https://doi.org/10.3390/vaccines12070766
Marissa E. Linger, Sytze H.T. Jorritsma, Jonna Bloeme-ter Horst, Jessika C. Zevenhoven-Dobbe, Finn Rijlaarsdam, Emil Colstrup, Macha Beijnes, Jutte J.C. de Vries, Ramon Arens, Rajagopal Murugan, Sebenzile K. Myeni, in collaboration with BREAK COVID group. Bivalent mRNA vaccine booster enhances immunity against XBB.1.5 more effectively than breakthrough infection in K18-hACE2 mice. iScience (2025). https://doi.org/10.1016/j.isci.2025.113479


